Basic paper - What is evaporation?
Content
1. What is a rotary evaporator?
A rotary evaporator is a laboratory device that allows the separation of two or more substances. This process is called distillation. To understand in detail what a rotary evaporator is and how it works, you must first understand the phenomenon of evaporation. This includes understanding the physical principle behind it.
2. What is evaporation?
Evaporation is a process that happens around us every day without us consciously noticing it:
- Laundry dries on the line because the water in the fabric evaporates.
- When water boils on the stove, steam rises from the pot.
Evaporation means: a liquid, for example water, becomes a gas. It changes its physical state (aggregate state) from liquid to gas. We also say: The substance changes from the liquid phase to the gaseous phase.
Let‘s stay with our pot of water: If we simply place the pot on the table, after a long time the water will seemingly disappear. But it is not simply gone: it has evaporated. So, little by little, the water goes from the liquid phase to the gaseous phase, without us contributing anything. To understand why this happens, we have to go to a level that the human eye cannot resolve. Let‘s take a closer look at the water in the pot with the help of Prof. Hei’s molecular magnifier glass:
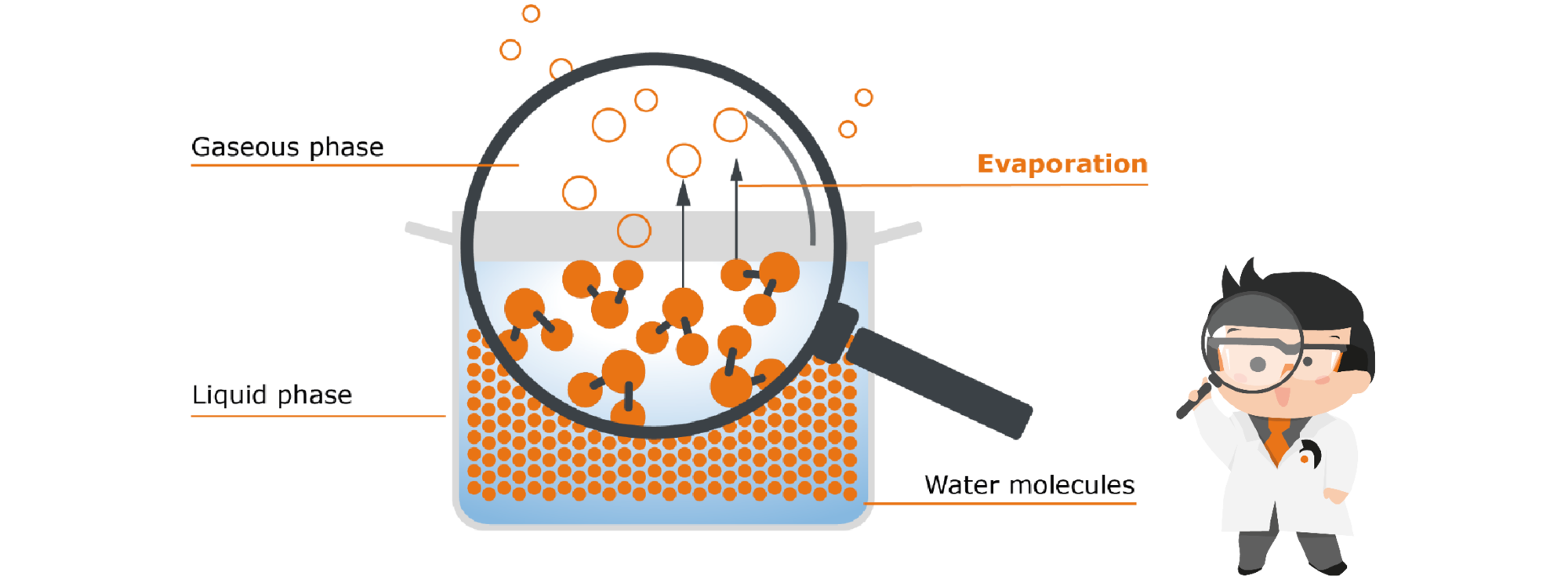
Fig. 1: Open pot with water
| In liquid water, the water molecules are very close to each other. They even hold on to each other by so-called intermolecular bonds, i.e. binding forces between the individual molecules. |  |
| Fig. 2: Water molecules with intermolecular bonds |
However, they do not simply lie static like marbles in the pot, but are constantly moving. These movements cause the molecules to collide with each other repeatedly, transferring kinetic energy from one molecule to the other. If a single molecule receives a particularly large number of these collisions, it obtains the necessary energy to break the bonds to its partner molecules - in other words, to free itself from the confines of the liquid and rise up out of it. This freedom is desirable for the molecule - understandable, isn‘t it? Who likes to be constricted? That is why molecules always strive to gain as much freedom as possible - they therefore pass from the liquid into the gas phase as soon as they get the necessary energy to do so.
The energy required to bring the molecules into the gas phase depends on their weight (molar weight) and the binding forces between them. This results in two physical quantities that are specific to each substance: The boiling point, which is the temperature at which a substance begins to boil (for water, this is 100 °C), and the enthalpy of vaporization. It indicates how much energy must be expended to evaporate one kilogram of the substance. For water, this value is 2261 kJ/kg, and others can be found in table 1.
|
|
Table 1: Molecular weights, boiling points and enthalpies of vaporization of some solvents
| Substance | Molecular weight [g/mol] |
Boiling point [°C] |
Enthalpy of vaporization [kJ/kg] |
| Toluene | 92.14 | 110 | 413 |
| Water | 18.02 | 100 | 2,261 |
| Ethanol | 46.07 | 78 | 837 |
| Acetone | 58.08 | 56 | 546 |
| Diethyl ether | 74.12 | 35 | 392 |
2.1 What happens when we place a pot of water on a hot plate and apply heat to it?
Heat is a form of energy. It causes our molecules to absorb this energy, making them much more agile. There is also an increase in the collisions of the molecules with each other. The result of all this is that the added heat gives many more molecules the energy they need to move from the liquid phase to the gaseous phase. More water therefore evaporates because of the heat supply. The maximum possible amount of vapor is produced when the boiling temperature is reached (i.e. 100 °C for water).
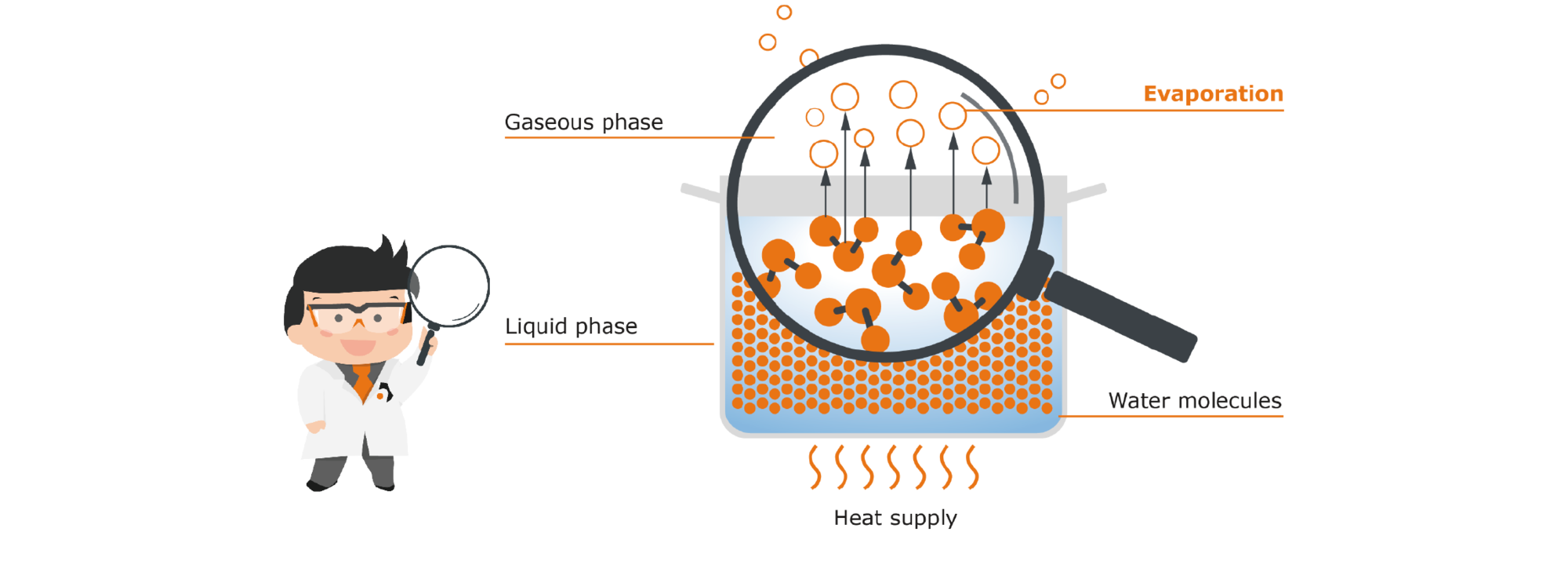
Fig. 3: Open pot with water supplied with heat
2.2 What is the influence of pressure?
Another variable we have not looked at so far is atmospheric pressure: Our pot of water, just like every-thing else, is subject to atmospheric pressure. This varies depending on the geographical altitude, but can be roughly given as 1,000 mbar. This pressure thus counteracts the transition of our water molecules from the liquid to the gas phase. Let us now imagine that we would reduce the atmospheric pressure, e.g. from 1,000 mbar to 50 mbar: Despite the same amount of energy supplied, considerably more of our molecules can now pass from the liquid phase into the gaseous phase.
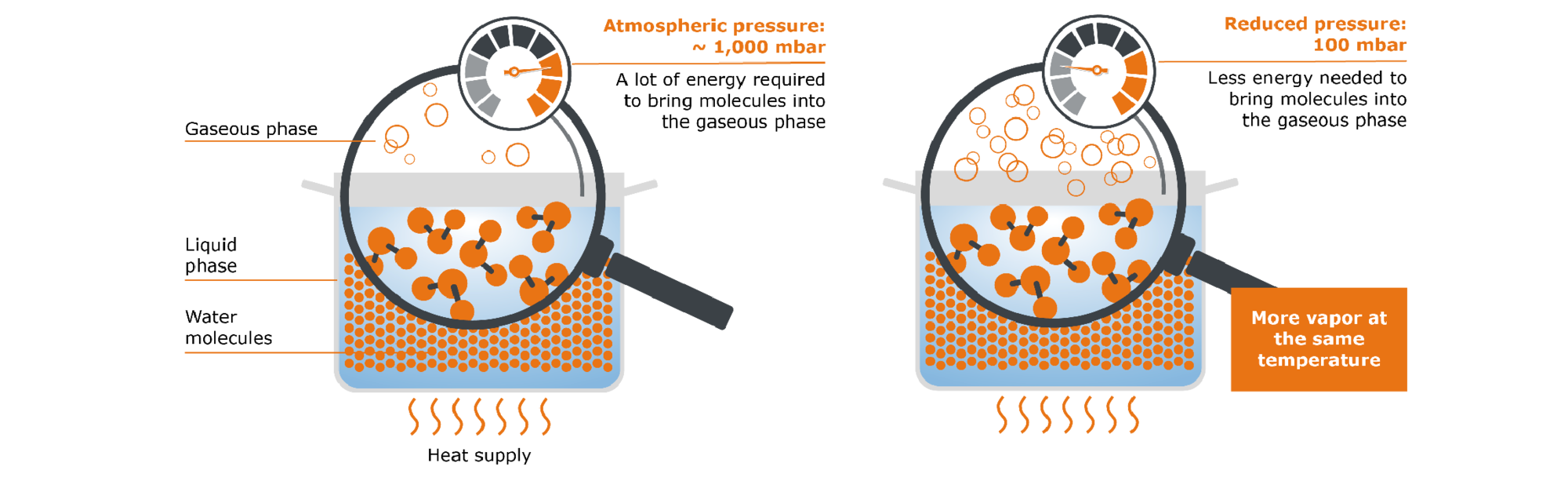
Fig. 4: Influence of pressure on open pot with water
A simple principle can be derived from this: The lower the pressure, the easier a substance can evaporate and the less heat is required for evaporation. The maximum possible amount of vapor can therefore already be generated at lower temperatures (e.g. 50 °C for water at 100 mbar).

3. What is condensation?
Condensation is, so to speak, the reverse process to evaporation: a substance passes from the gaseous phase to the liquid phase. This happens when the energy we have supplied to our liberated water molecules is taken away again. The easiest way to do this is through cold.
To approach the phenomenon of condensation, we again use our example of the pot with water: If we now close the pot with a lid, the steam can no longer simply escape, but it will remain in this closed system of the pot interior. At the same time, the lid of the pot is cooler than the rest: our liberated water molecules therefore collide with this cool barrier and transfer some of their energy to the cooler lid. A temperature equalization takes place. The molecules collect on the cold surface to form droplets. They condense, which is why the condensed water is called ‚condensate‘. At some point, the drops are heavy enough and fall back into the liquid, where the molecules are re-energized and can rise again.
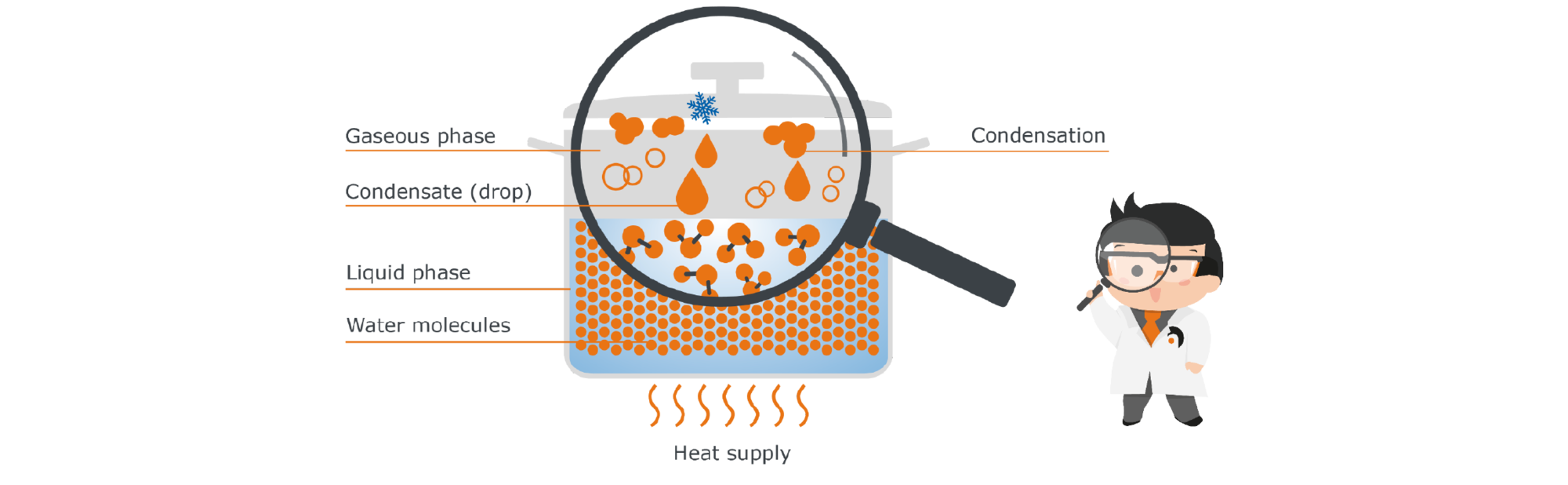
Fig. 5: Condensation inside a closed pot
| A cycle of evaporation and condensation is formed, which is in equilibrium. | 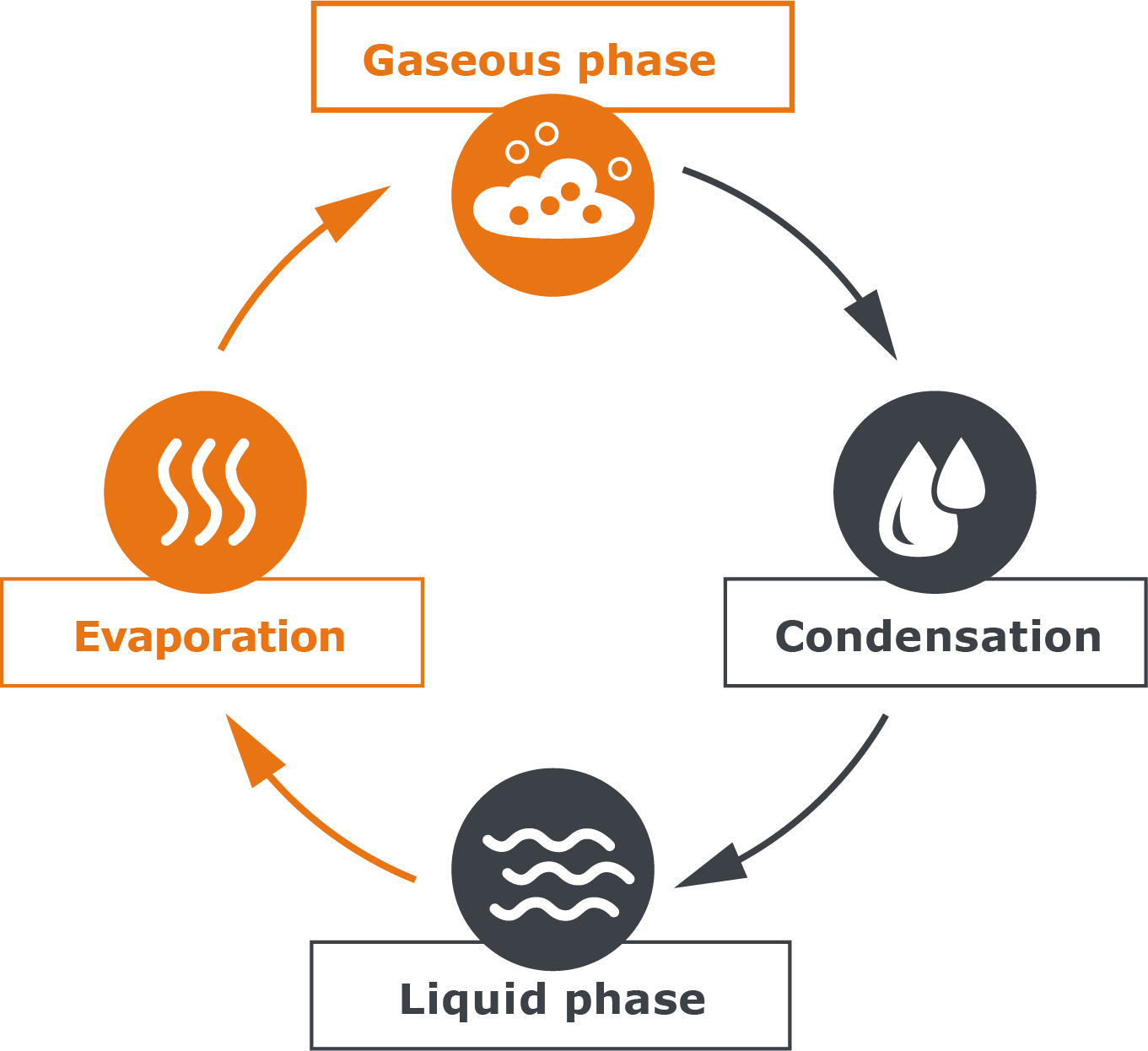 |
| Fig. 6: Equilibrium between evaporation and condensation |
4. What is distillation?
A distillation is a separation of substances with different boiling points. Here, first the substance with the lowest boiling point is evaporated and condensed again at another point, then by increasing the temperature the next substance and so on. In the laboratory, distillation is an important method for substance separation or purification.
A classic apparatus for this process is the static still:
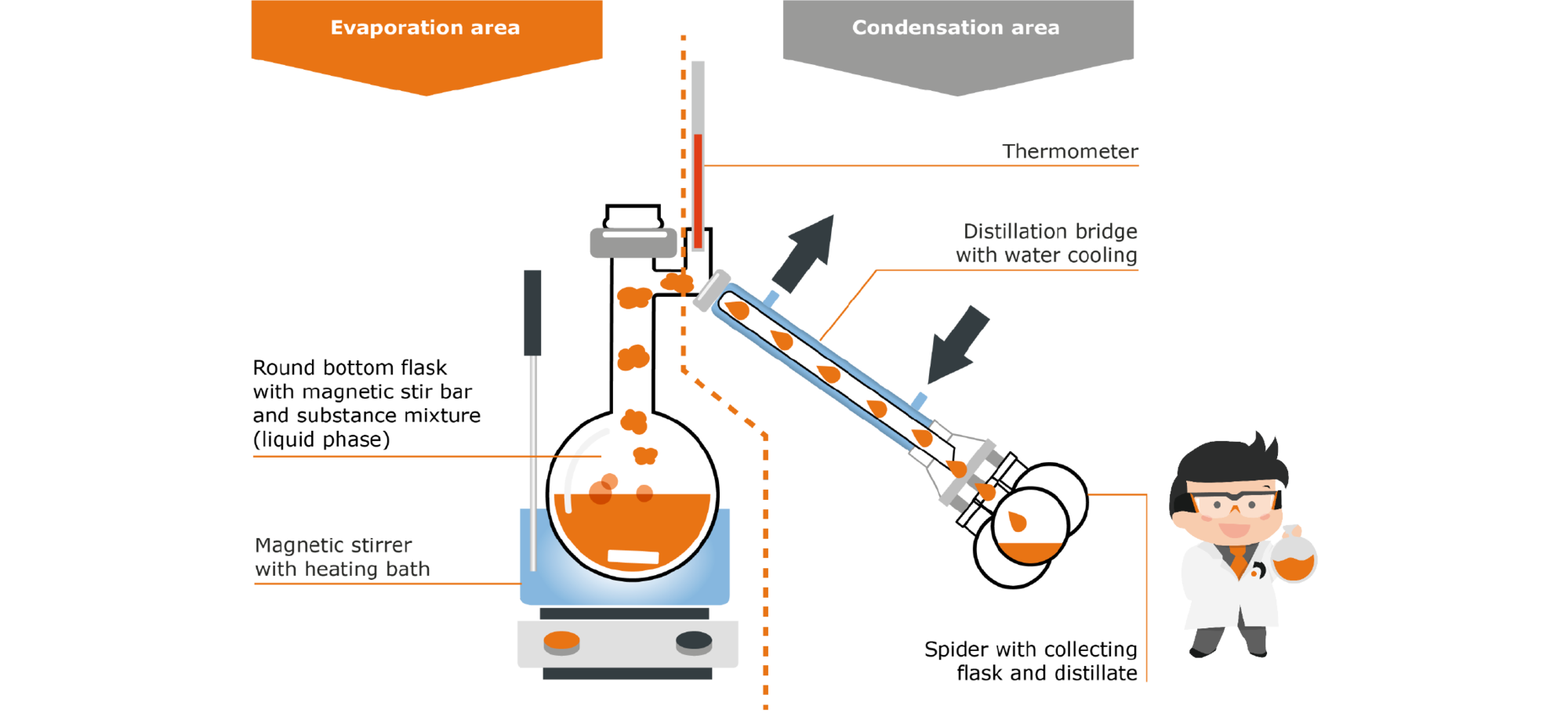
Fig. 7: Set-up a static still
You can roughly divide the apparatus into two sections: The evaporation area and the condensation area. In the evaporation area, the mixture of substances is heated in a flask in a heating bath. The vapor of the substance with the lowest boiling point rises to the top and is led across to the condensation area in the so-called distillation bridge. It is cooled with water, which leads to condensation of the vapor. The droplets run down due to the inclination of the bridge and drip into the collecting flask. Once the first component (fraction) has been completely separated, the so-called spider can simply be rotated so that the next free collecting flask points downwards and collects the distillate. The temperature at the heating bath is increased until the boiling point of the next component (second fraction) is reached. At the end of the process, the separated substances are cleanly present in the individual collecting flasks.

Fig. 8: Distillation process with static still
Distillation can also be carried out under reduced pressure by connecting the still to a vacuum pump: This allows work to be carried out at milder temperatures, which is particularly advantageous for heat-sensitive substances.
5.What is the difference between a static still and a rotary evaporator?
The rotary evaporator is also a laboratory device used for the distillate separation of substances. Thus, the rotary evaporator design can also be divided into the two areas of evaporation and condensation.
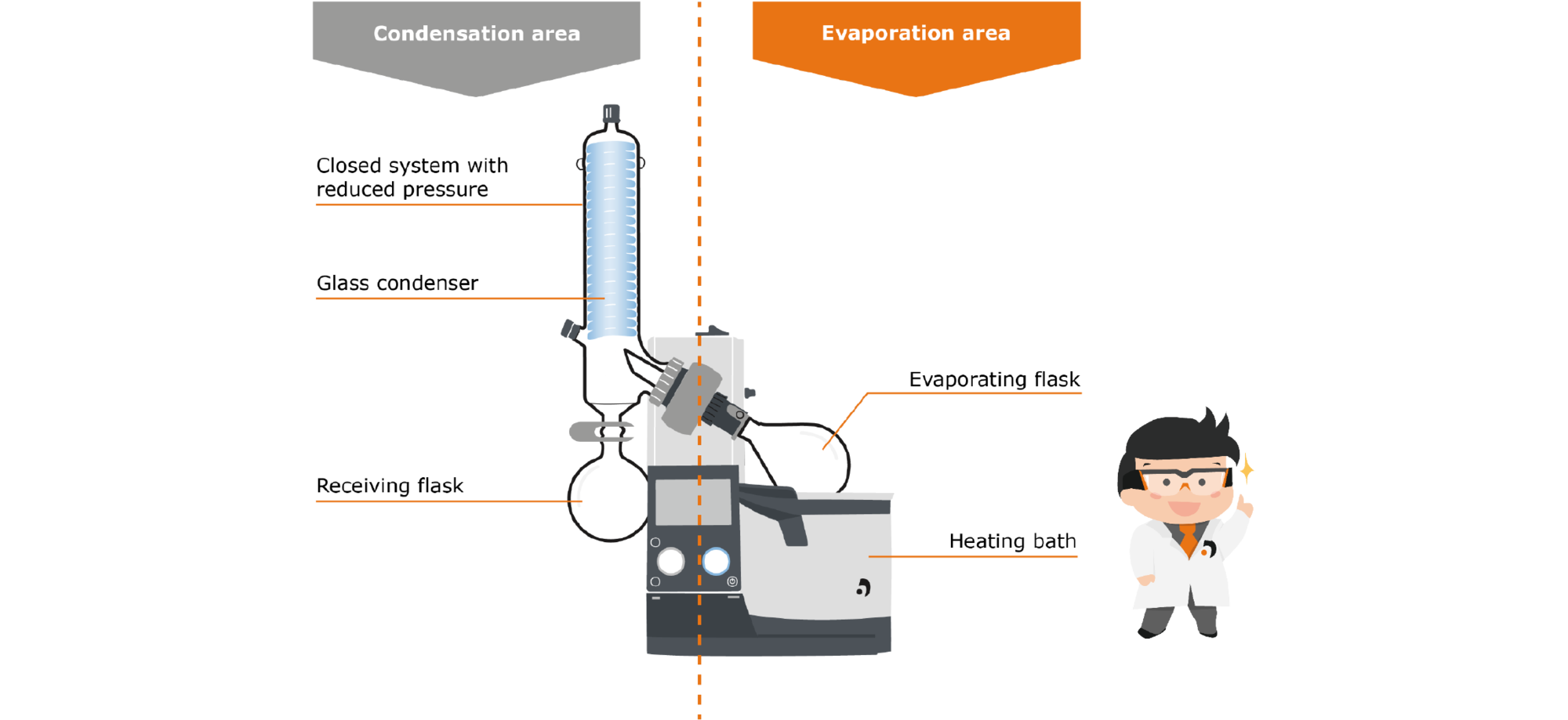
Fig. 9: Set-up a rotary evaporator
First, let‘s take a closer look at the evaporation area: The area includes the evaporator flask, which contains the liquid medium. This flask is immersed in the heating bath, which supplies the energy necessary for evaporation. Unlike the static still, however, the flask is set in rotation by a motor. This has several effects: First, the temperature distribution in the heating bath and inside the flask takes place more uniformly, and second, the rotation significantly increases the surface area available for evaporation. This has a positive effect on the evaporation speed as well as on the formation of bubbles and spills.
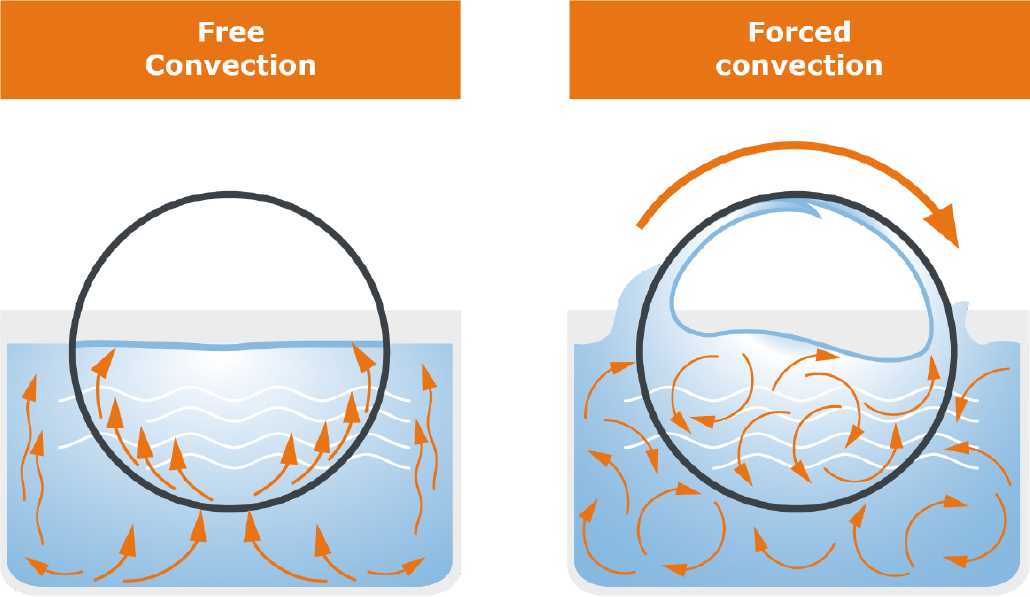
Fig. 10: Static system compared to rotating system
The generated steam rises through the steam guide tube into the condensation zone: Here there is a glass cooler cooled either with a dry ice cooling mixture, water or a coolant, on which the steam condenses and drips down into the receiving flask.
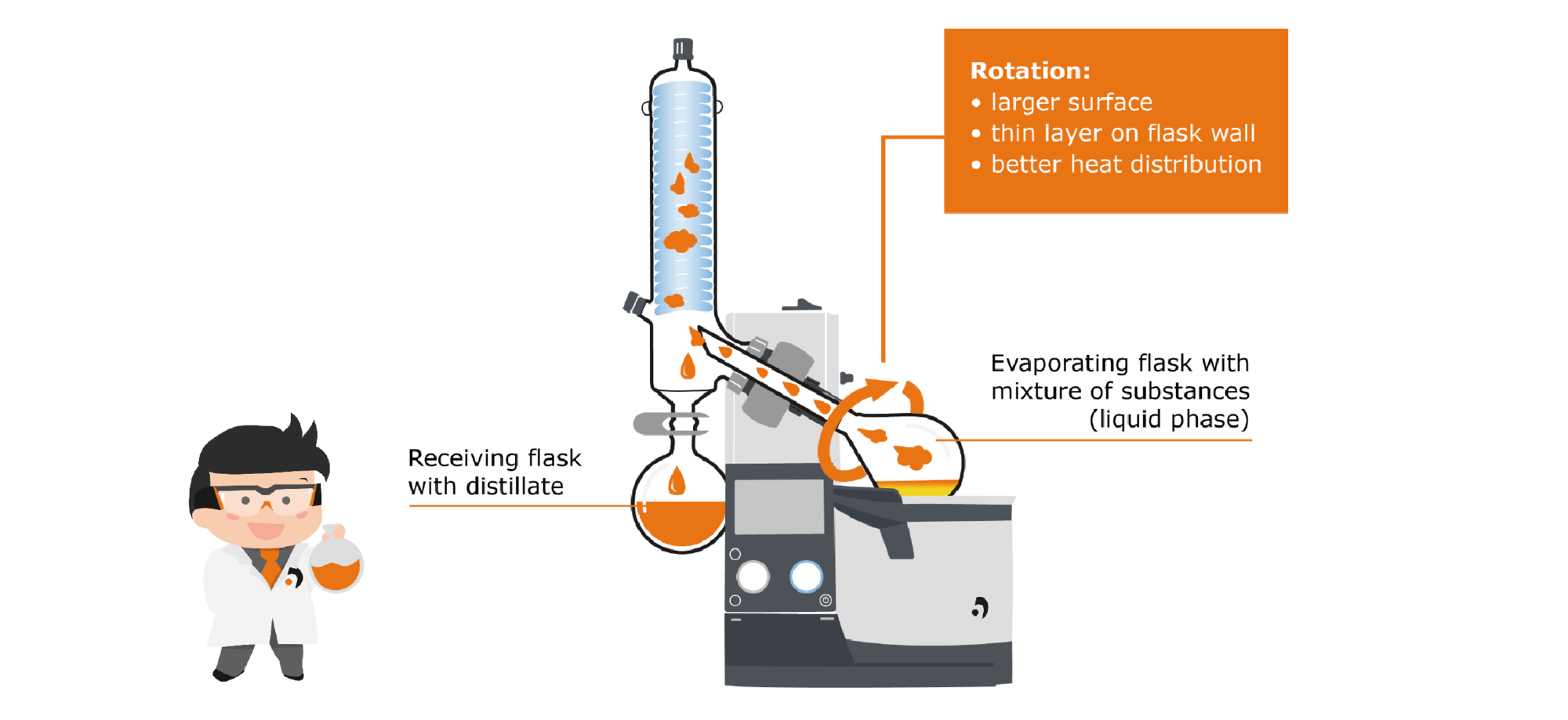
Fig. 11: Evaporation process on rotary evaporator
The whole process is usually carried out under pressure reduced by a vacuum pump to take advantage of the benefits already mentioned above: A lower energy input is necessary to trigger the evaporation and thus the materials are processed more gently. In addition, working with a vacuum pump gives one access to a very quickly adjustable parameter to influence the process speed, since no long maintenance times, such as those caused by the inertia of a heating bath, are required.
The separation performance of a rotary evaporator is relatively low due to the short distance the vapor has to travel: to achieve a clean separation of substances, the boiling points of the substances to be separated should be at least 80 °C apart.
Downloads
|
|